A) linear.
B) trigonal planar.
C) tetrahedral.
D) bent.
E) trigonal pyramidal.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the hybridization for the O in OF2.
A) sp
B) sp3
C) sp2
D) sp3d
E) sp3d2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the number of electron groups around a molecule with sp3d2 hybridization.
A) 6
B) 2
C) 3
D) 4
E) 5
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule,that is sp3d hybridized and has a molecular geometry of seesaw,has ________ bonding groups and ________ lone pairs around its central atom.
A) 5, 1
B) 4, 2
C) 4, 1
D) 3, 2
E) 2, 3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What geometric arrangement of charge clouds is expected for an atom that has three charge clouds?
A) trigonal bipyramidal
B) linear
C) trigonal
D) tetrahedral
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Draw the molecular orbital diagram shown to determine which of the following is most stable.
A) F2
B) F22⁺
C) Ne22⁺
D) O22⁺
E) F22⁻
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the molecular orbital diagram shown to determine which of the following is paramagnetic. 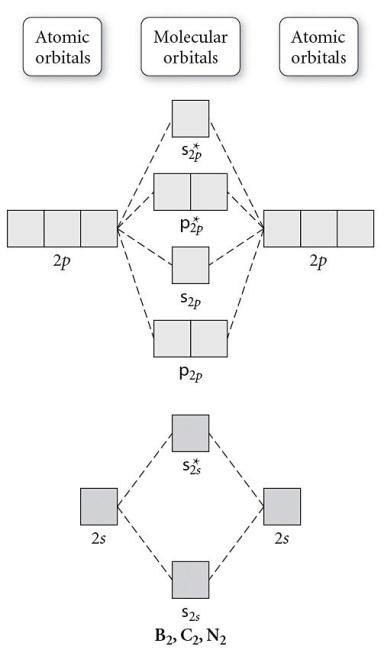
A) B22⁺
B) B22⁻
C) N22⁺
D) C22⁻
E) B2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule containing a central atom with sp3d hybridization has a(n) ________ electron geometry.
A) seesaw
B) bent
C) octahedral
D) trigonal planar
E) trigonal bipyramidal
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule,that is sp3d2 hybridized and has a molecular geometry of square pyramidal,has ________ bonding groups and ________ lone pairs around its central atom.
A) 5, 1
B) 4, 2
C) 4, 1
D) 3, 2
E) 2, 3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Place the following in order of decreasing X-A-X bond angle,where A represents the central atom and X represents the outer atoms in each molecule. CS2 CF4 SCl2
A) CS2 = SCl2 > CF4
B) SCl2 > CF4 > CS2
C) CF4 > CS2 > SCl2
D) CS2 > CF4 > SCl2
E) CF4 > CS2 > SCl2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the number of electron groups around a molecule with sp3d hybridization.
A) 1
B) 2
C) 3
D) 4
E) 5
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Draw the Lewis structure for the molecule CH2CHCH3.How many sigma and pi bonds does it contain?
A) 8 sigma, 1 pi
B) 9 sigma, 0 pi
C) 9 sigma, 1 pi
D) 7 sigma, 0 pi
E) 8 sigma, 2 pi
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule containing a central atom with sp hybridization has a ________ electron geometry.
A) linear
B) trigonal bipyramidal
C) trigonal planar
D) octahedral
E) bent
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the O-S-O bond angle in SO3?
A) 45°
B) 109.5°
C) 120°
D) 210°
E) 107°
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the electron geometry (eg) ,molecular geometry (mg) ,and hybridization for NCl3.
A) eg = tetrahedral, mg = trigonal planar, sp2
B) eg = trigonal pyramidal, mg = trigonal pyramidal, sp3
C) eg = tetrahedral, mg = trigonal pyramidal, sp3
D) eg = trigonal pyramidal, mg = tetrahedral, sp3
E) eg = trigonal planar, mg = trigonal planar, sp2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the electron geometry (eg) ,molecular geometry (mg) ,and hybridization for H2Se.
A) eg = tetrahedral, mg = bent, sp3
B) eg = trigonal pyramidal, mg = trigonal planar, sp3
C) eg = tetrahedral, mg = trigonal pyramidal, sp3
D) eg = bent, mg = tetrahedral, sp2
E) eg = tetrahedral, mg = tetrahedral, sp2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the electron geometry (eg) ,molecular geometry (mg) ,and polarity of SO3.
A) eg = tetrahedral, mg = trigonal pyramidal, polar
B) eg = tetrahedral, mg = tetrahedral, nonpolar
C) eg = trigonal planar, mg = trigonal planar, nonpolar
D) eg = trigonal bipyramidal, mg = trigonal pyramidal, nonpolar
E) eg = trigonal pyramidal, mg = bent, nonpolar
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the electron geometry (eg) and molecular geometry (mg) of HCN.
A) eg = tetrahedral, mg = tetrahedral
B) eg = linear, mg = trigonal planar
C) eg = trigonal planar, mg = bent
D) eg = linear, mg = linear
E) eg = trigonal planar, mg = trigonal planar
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following compound.How many sigma and pi bonds does it contain? CH3CHCHCO2H
A) 9 sigma, 4 pi
B) 11 sigma, 2 pi
C) 9 sigma, 0 pi
D) 13 sigma, 2 pi
E) 6 sigma, 2 pi
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the electron geometry (eg) and molecular geometry (mg) of the underlined carbon in CH3CN.
A) eg = tetrahedral, mg = tetrahedral
B) eg = linear, mg = bent
C) eg = trigonal planar, mg = linear
D) eg = linear, mg = linear
E) eg = bent, mg = tetrahedral
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 144
Related Exams