A) the change in density when it condenses to form a liquid or solid
B) reactions with other atmospheric compounds
C) the release of heat by the formation of hydrogen bonds
D) the release of heat by the breaking of hydrogen bonds
E) the high surface tension of water
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One of the buffers that contribute to pH stability in human blood is carbonic acid H₂CO₃. Carbonic acid is a weak acid that when placed in an aqueous solution dissociates into a bicarbonate ion (HCO₃⁻) and a hydrogen ion (H⁺) . Thus, H₂CO₃ ↔ HCO₃⁻ + H⁺ If the pH of the blood increases, one would expect
A) a decrease in the concentration of H₂CO₃ and an increase in the concentration of H₂O.
B) an increase in the concentration of H₂CO₃ and a decrease in the concentration of H₂O.
C) a decrease in the concentration of HCO₃⁻ and an increase in the concentration of H₂O.
D) an increase in the concentration of HCO₃⁻ and a decrease in the concentration of H₂O.
E) a decrease in the concentration of HCO₃⁻ and an increase in the concentration of both H₂CO₃ and H₂O.
G) None of the above
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
One mole (mol) of a substance is
A) 6) 02 × 10²³ molecules of the substance.
B) 1 g of the substance dissolved in 1 L of solution.
C) the largest amount of the substance that can be dissolved in 1 L of solution.
D) the molecular mass of the substance expressed in grams.
E) A and D only
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which type of bond must be broken for water to vaporize?
A) ionic bonds
B) nonpolar covalent bonds
C) polar covalent bonds
D) hydrogen bonds
E) covalent bonds
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ionizes completely in solution and is considered to be a strong base (alkali) ?
A) NaCl
B) HCl
C) NH₃
D) H₂CO₃
E) NaOH
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A slice of pizza has 500 kcal. If we could burn the pizza and use all the heat to warm a 50-L container of cold water, what would be the approximate increase in the temperature of the water? (Note: A liter of cold water weighs about 1 kg.)
A) 50°C
B) 5°C
C) 10°C
D) 100°C
E) 1°C
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One of the buffers that contribute to pH stability in human blood is carbonic acid (H₂CO₃) . Carbonic acid is a weak acid that dissociates into a bicarbonate ion (HCO₃⁻) and a hydrogen ion (H⁺) . Thus, H₂CO₃ ↔ HCO₃⁻ + H⁺ If the pH of the blood drops, one would expect
A) a decrease in the concentration of H₂CO₃ and an increase in the concentration of HCO₃⁻.
B) the concentration of hydroxide ion (OH⁻) to increase.
C) the concentration of bicarbonate ion (HCO₃⁻) to increase.
D) the HCO₃⁻ to act as a base and remove excess H⁺ with the formation of H₂CO₃.
E) the HCO₃⁻ to act as an acid and remove excess H⁺ with the formation of H₂CO₃.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution contains 0.0000001(10⁻⁷) moles of hydroxyl ions [OH⁻] per liter. Which of the following best describes this solution?
A) acidic: H⁺ acceptor
B) basic: H⁺ acceptor
C) acidic: H⁺ donor
D) basic: H⁺ donor
E) neutral
G) B) and E)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
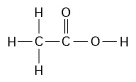 -How many grams of the molecule in Figure 3.2 would be equal to 1 mol of the molecule?
(Carbon = 12, Oxygen = 16, Hydrogen = 1)
-How many grams of the molecule in Figure 3.2 would be equal to 1 mol of the molecule?
(Carbon = 12, Oxygen = 16, Hydrogen = 1)
A) 29
B) 30
C) 60
D) 150
E) 342
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An example of a hydrogen bond is the bond between
A) C and H in methane (CH₄) .
B) the H of one water molecule and the O of another water molecule.
C) Na⁺and Cl⁻ in salt.
D) the two hydrogen atoms in a molecule of hydrogen gas (H₂) .
E) Mg⁺and Cl⁻ in MgCl₂.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 100 mL beaker contains 10 mL of NaOH solution at pH = 13. A technician carefully pours into the beaker, 10 mL of HCl at pH = 1. Which of the following statements correctly describes the results of this mixing?
A) The concentration of Na⁺ ion rises.
B) The concentration of Cl⁻ ion falls.
C) The concentration of undissociated H₂O molecules remains unchanged.
D) The pH of the beaker's contents rises.
E) The pH of the beaker's contents falls.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbon dioxide (CO₂) is readily soluble in water, according to the equation CO₂ + H₂O → H₂CO₃. Carbonic acid (H₂CO₃) is a weak acid. If CO₂ is bubbled into a beaker containing pure, freshly-distilled water, which of the following graphs correctly describes the results?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
 -How many grams of the molecule in Figure 3.2 would be required to make 2.5 L of a 1 M solution of the molecule?
(Carbon = 12, Oxygen = 16, Hydrogen = 1)
-How many grams of the molecule in Figure 3.2 would be required to make 2.5 L of a 1 M solution of the molecule?
(Carbon = 12, Oxygen = 16, Hydrogen = 1)
A) 29
B) 30
C) 60
D) 150
E) 342
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assume that acid rain has lowered the pH of a particular lake to pH 4.0. What is the hydroxyl ion concentration of this lake?
A) 1 × 10⁻¹⁰ mol of hydroxyl ion per liter of lake water
B) 1 × 10⁻⁴ mol of hydroxyl ion per liter of lake water
C) 10.0 M with regard to hydroxyl ion concentration
D) 4.0 M with regard to hydroxyl ion concentration
E) both B and D
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At what temperature is water at its densest?
A) 0°C
B) 4°C
C) 32°C
D) 100°C
E) 212°C
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When an ionic compound such as sodium chloride (NaCl) is placed in water the component atoms of the NaCl crystal dissociate into individual sodium ions (Na⁺) and chloride ions (Cl⁻) . In contrast, the atoms of covalently bonded molecules (e.g., glucose, sucrose, glycerol) do not generally dissociate when placed in aqueous solution. Which of the following solutions would be expected to contain the greatest number of particles (molecules or ions) ?
A) 1 L of 0.5 M NaCl
B) 1 L of 0.5 M glucose
C) 1 L of 1.0 M NaCl
D) 1 L of 1.0 M glucose
E) C and D will contain equal numbers of particles.
G) C) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the hydroxide ion concentration of the lake described in question 7?
A) 10⁻⁷ M
B) 10⁻⁴ M
C) 10⁻¹⁰ M
D) 10⁻¹⁴ M
E) 10 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
We can be sure that a mole of table sugar and a mole of vitamin C are equal in their
A) mass in daltons.
B) mass in grams.
C) number of molecules.
D) number of atoms.
E) volume.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a solution with a hydroxyl ion [OH⁻] concentration of 10⁻¹² M?
A) pH 2
B) pH 4
C) pH 10
D) pH 12
E) pH 14
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of acetic acid (C₂H₄O₂) would you use to make 10 L of a 0.1 M aqueous solution of acetic acid? (Note: The atomic masses, in daltons, are approximately 12 for carbon, 1 for hydrogen, and 16 for oxygen.)
A) 10.0 g
B) 0.1 g
C) 6.0 g
D) 60.0 g
E) 0.6 g
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 66
Related Exams