A) They are all halogens.
B) They all have the same electron configuration.
C) They are all nonmetals.
D) They are all representative elements.
E) They all have the same number of valence electrons.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Where are the halogens located on the periodic table?
A) representative elements
B) transition metals
C) Group VIIA (17)
D) Group IIA (2)
E) Group IIIA (3)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many orbitals are there in the second principal energy level?
A) 2
B) 3
C) 1
D) 0
E) 4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The last period in the periodic table is incomplete.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is a metalloid?
A) C
B) Ge
C) Pb
D) N
E) P
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The element carbon forms the basis of study in Organic Chemistry.Which statement about the element carbon is FALSE?
A) Carbon is a period 2 element.
B) Carbon is a group 4 element.
C) Carbon is a nonmetal.
D) Carbon atoms have six valence electrons.
E) Carbon atoms have six protons.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When is an atom in its ground state?
A) when the electrons of the atom are in the lowest possible energy levels
B) when an atom loses all of its electrons to form a noble gas
C) when the electrons of the atom are in the highest possible energy levels
D) when an electron is promoted to a higher energy level, farther from the nucleus
E) when the electrons of an atom spontaneously emit energy in the form of a photon
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rutherford's experiment, in which alpha particles were aimed at a thin piece of gold, led to what understanding?
A) Neutrons existed.
B) Electrons existed and have a negative charge.
C) The number of electrons can be determined by the mass number and atomic number.
D) Electrons can be promoted to higher energy by absorbing energy.
E) An atom is mostly empty space.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The identity of an atom is determined by which of the following?
A) the number of electrons it contains
B) its mass number
C) the number of isotopes it has
D) the number of protons it contains
E) the number of protons and neutrons it contains
G) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of the following elements has the lowest ionization energy?
A) Li
B) B
C) O
D) F
E) Ne
G) B) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
How many neutrons are present in an atom of the isotope 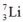 ?
?
A) 3
B) 4
C) 7
D) 10
E) None of the choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atoms with the biggest radii occur in the _______ region of the periodic table.
A) bottom left
B) top right
C) bottom right
D) top left
E) middle
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The atoms of smallest radius are those of elements in the top left hand part of the periodic table.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the highest ionization energy?
A) Li
B) B
C) O
D) F
E) He
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many orbitals are in an s sublevel? How many in a p sublevel?
A) s: 1, p: 2
B) s: 2, p: 3
C) s: 1, p: 3
D) s: 2, p: 6
E) s: 3, p: 3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Microwaves, light, and X-rays are all forms of
A) electricity.
B) high energy electrons.
C) electron repulsion.
D) electromagnetic radiation.
E) radioactivity.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the lowest energy sublevel of a principal level?
A) d
B) e
C) f
D) s
E) p
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In Mendeleev's table of the elements, the elements were arranged according to
A) atomic number.
B) mass number.
C) atomic mass.
D) neutron number.
E) density.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many sublevels are there in the third principal energy level?
A) 3
B) 2
C) 1
D) 0
E) 4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Arsenic (As) is a metalloid.
B) False
Correct Answer

verified
True
Correct Answer
verified
Showing 1 - 20 of 109
Related Exams